 \(\pi\) bonds are created when there is adequate overlap of similar, adjacent \(p\) orbitals, such as \(p_x\) \(p_x\) and \(p_y\) \(p_y\). Common sigma bonds are \(s s\), \(p_z p_z\) and \(s p_z\), \(z\) is the axis of the bond on the xyz-plane of the atom. Sigma bonds are created when there is overlap of similar orbitals, orbitals that are aligned along the inter-nuclear axis. As opposed to ionic bonds which hold atoms together through the attraction of two ions of opposite charges. When this photon hits the carbon atom it gives the atom enough energy to promote one of the lone pair electrons to the \(2p_z\) orbital.Īll the bonds in Ethene are covalent, meaning that they are all formed by two adjacent atoms sharing their valence electrons. It becomes promoted when a photon of light with the correct wavelength hits the carbon atom. The electron is not promoted spontaneously. Well it is, in order to make the four bonds, the carbon atom promotes one of the 2s electrons into the empty \(2p_z\) orbital, leaving the carbon with four unpaired electrons allowing it to now form four bonds. 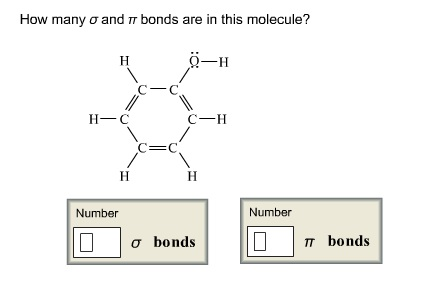
The other two are in a lone pair state, making them much less reactive to another electron that is by itself. A single carbon atom can make up to four bonds, but by looking at its electron configuration this would not be possible because there are only two electrons available to bond with. With four single bonds, carbon has a tetrahedral structure, while with one double bond it's structure is trigonal planar, and with a triple bond it has a linear structure.Ī solitary carbon atom has four electrons, two in the 2s orbital, and one in each of the 2\(p_x\) and 2\(p_y\) orbitals, leaving the \(2p_z\) orbital empty. The number of bonds it makes determines the structure. Carbon can make single, double, or triple bonds. Each carbon has 4 and each hydrogen 1 for a total of 10 electrons.\)īonding in carbon is covalent, containing either sigma or \(\pi\) bonds. Add the valence electrons to the molecular orbital diagram.The 2p y orbitals on each carbon combine to make another 2 pi symmetry orbitals, 90 degrees from the first set. The 2p x orbitals on each atom combine to make 2 pi symmetry orbitals.(C-H bonds)Ĭombine the other 2 C(2sp) orbitals to make a sigma bonding and a sigma antibonding molecular orbital. Combine each H(1s) orbital with a C(2sp) orbital to make a sigma bonding and a sigma antibonding molecular orbital. 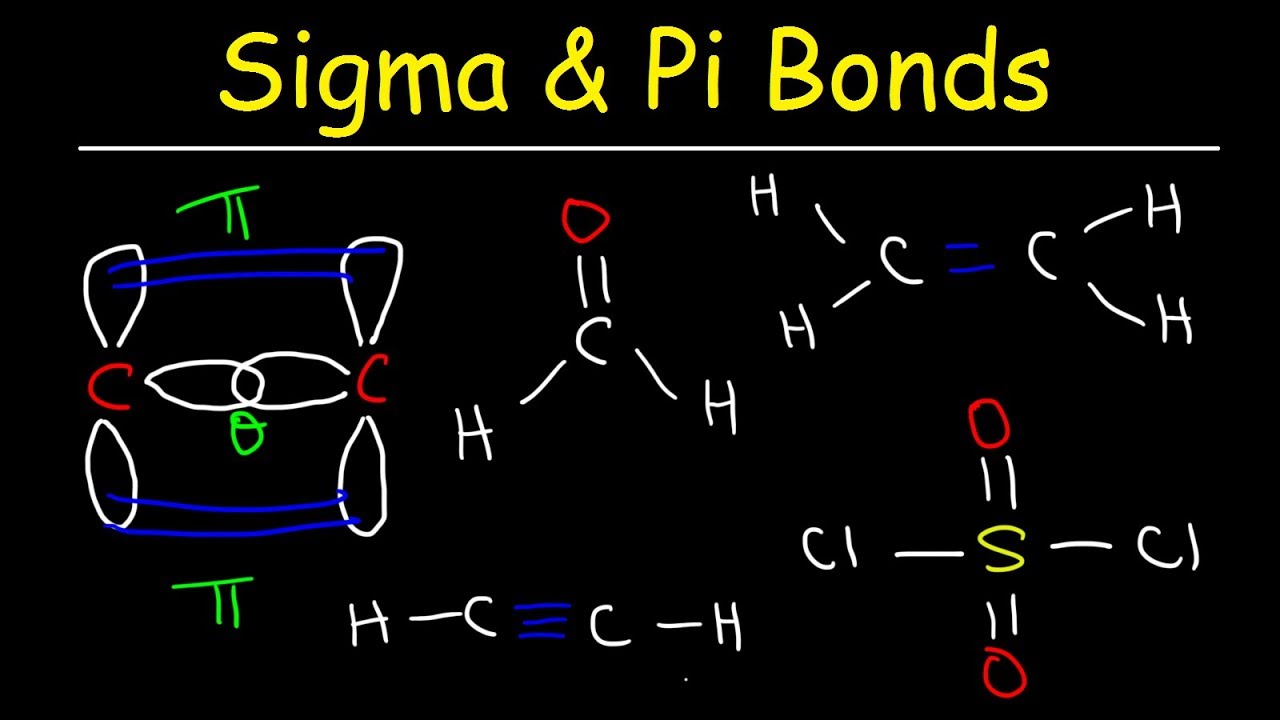
After hybridization, a 2p x and a 2p y orbital remain on each carbon atom. The carbon atoms in ethyne use 2sp hybrid orbitals to make their sigma bonds.Each carbon atom makes 2 sigma bonds and has no lone pairs of electrons. Each carbon has 4 and each hydrogen 1 for a total of 12 electrons.Įthyne, sp hybridization with two pi bonds Finally, add the valence electrons to the molecular orbital diagram.The stabilization and destabilization in forming a pi bond are much less than for a sigma bond. The stabilization (decrease in energy) in going from the p orbital to pi bonding orbital equals the destabilization (increase in energy) in going from the p orbital to the pi antibonding orbital. These can combine to make a pi bonding and a pi antibonding molecular orbital. There remains a 2p orbital on each carbon. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
